The Ultimate Guide to Grains: Myths, Facts & The Microbiome Connection
Introduction: The Great Grain Debate – Rethinking the Role of Grains in Gut Health
For years, grains have been demonised in health and wellness circles, with claims that they inflame the gut, trigger weight gain, and wreak havoc on the microbiome. Many practitioners—even some of the best in the field—have advised patients to avoid grains altogether, often based on early research or oversimplified interpretations of the science.
But what if we’ve been misled? What if the problem isn’t the grains themselves but how we approach them in clinical practice?
The truth is, that grains are not inherently harmful. In fact, they play a crucial role in microbial diversity, gut barrier integrity, and metabolic health when consumed in the right way. The challenge isn’t whether grains should be in the diet—it’s about understanding which grains, for whom, and in what form.
As a practitioner, your job is not to blindly follow diet trends but to critically assess the evidence and tailor recommendations based on individual needs. Are grains really the enemy, or have we overlooked their benefits in the rush to vilify gluten? Are all grains created equal, or is it time to distinguish between refined, processed grains and whole, fiber-rich varieties?
This article is designed to challenge the common misconceptions around grains and provide you with a clear, evidence-based framework for integrating them into clinical practice. By the end, you’ll have the confidence to determine when grains are appropriate, how to introduce them safely, and how to support your patients in making informed dietary choices.
Because the best practitioners don’t just follow the science—they grow with it.
Popular Myths and Misconceptions About Grains
Grains have been at the center of heated debates in nutrition for years. Some claim they destroy gut health, others blame them for weight gain and inflammation, and gluten has practically become a villain in the wellness world. But what if much of what we’ve been told is based on misinterpretations of the science?
Let’s break down the most common myths and see what the evidence actually says.
-
For years, gluten has been accused of increasing intestinal permeability (aka “leaky gut”) in everyone, not just those with coeliac disease. Early studies showed that gluten exposure could increase gut permeability markers, which sparked widespread fear.
But here’s the truth: The research that linked gluten to leaky gut was largely based on in vitro (lab) studies where it can't be translated to human studies. In healthy individuals, there is no strong evidence that gluten significantly disrupts the intestinal barrier. However, in HLA-DQ2/8-positive patients, when gluten is consumed, there is intestinal permeability.
✅ Clinical Pearl: Instead of assuming all patients need to avoid gluten, test for markers of gut permeability (e.g., stool zonulin, Hexa-LPS and butyrate) before making sweeping dietary restrictions.
-
Many practitioners believe that grains harm the gut microbiome, but the reality is far more nuanced. The type of grain—and how it’s processed—matters more than the grain itself.
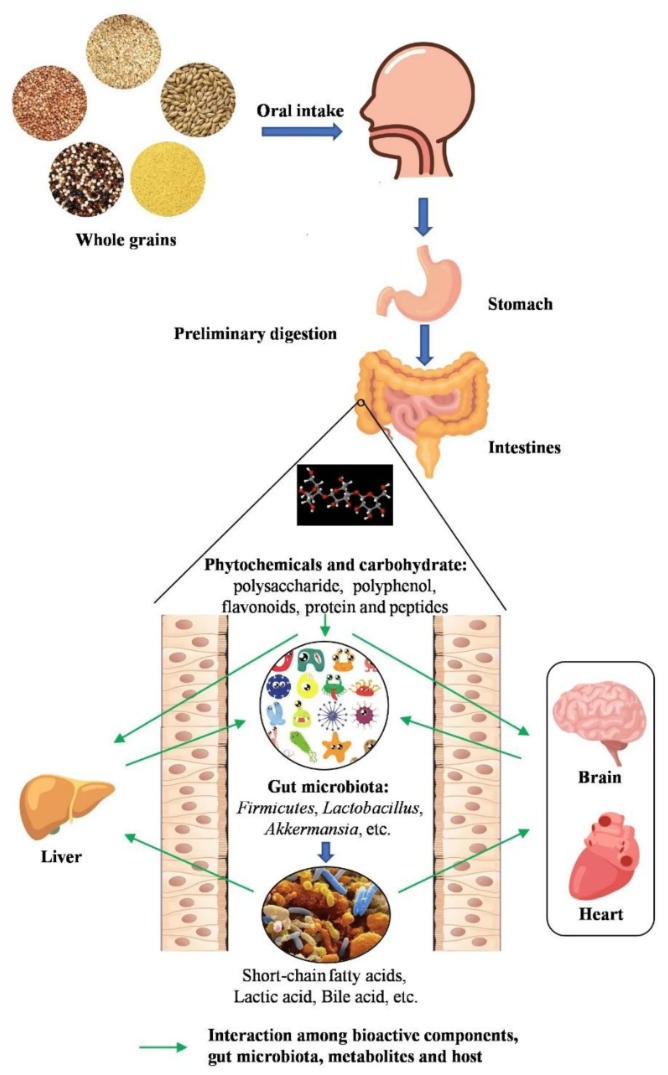
Refined, processed grains (think white bread, sugary cereals, and instant noodles) do little for gut health. But whole grains like oats, rye, barley, and quinoa are rich in fibre, resistant starch, and polyphenols that nourish beneficial gut bacteria.
Studies show that fibre from whole grains increases short-chain fatty acid (SCFA) production, particularly butyrate, which supports gut barrier function, reduces inflammation, and promotes microbial diversity. In contrast, low-fibre diets starve beneficial bacteria and may even lead to the overgrowth of mucus-degrading microbes that compromise gut integrity.
✅ Clinical Pearl: Encourage patients to focus on whole, minimally processed grains rather than simply labelling all grains as “bad.”
-
The idea that grains inherently cause inflammation has been widely repeated—but where’s the proof?
Most studies linking grains to inflammation focus on processed, high-glycemic grain products that spike blood sugar and contribute to metabolic dysfunction. Whole grains, however, are consistently associated with lower markers of inflammation, improved blood lipid profiles, and better metabolic health.
One study even found that whole grain rye consumption reduced C-reactive protein (CRP), a key marker of systemic inflammation. Instead of blaming grains for inflammation, we should be questioning which grains and how they’re consumed.
✅ Clinical Pearl: If a patient reports feeling inflamed after eating grains, investigate other dietary factors, such as ultra-processed foods, poor gut microbiome diversity, or even food preparation methods, before assuming grains are the culprit.
-
The “ancestral diet” argument claims that because early humans didn’t consume grains, they must be unnatural for our digestive system. But archaeology and genetics tell a different story.
Evidence suggests that humans have been eating grains for at least 100,000 years—long before the agricultural revolution. Ancient grinding tools show that early humans processed grains for consumption, and genetic adaptations (such as increased amylase enzyme production) indicate that our bodies have evolved to digest starchy foods efficiently.
✅ Clinical Pearl: Rather than dismissing grains based on evolutionary assumptions, consider how traditional grain preparation techniques like soaking, sprouting, and fermenting improve digestibility and nutrient absorption.
Understanding the Impact of Grains on Gut Health
Grains have been wrongly accused of being gut-destroying, inflammation-triggering, and microbiome-wrecking foods. The truth? It’s not grains that are the problem—it’s how they’re consumed, processed, and misunderstood.
For too long, we’ve lumped all grains together, ignoring the key differences between ultra-processed white bread and nutrient-dense whole grains like rye, barley, and quinoa. It’s time to separate fact from fiction and explore how grains—when chosen wisely—can actually support gut health.
Grains as Fuel for the Microbiome
If you want to improve gut health, you have to feed the microbiome. But here’s the problem: most people are starving their gut bacteria.
A diet rich in diverse, fermentable fibres—like those found in whole grains—promotes microbial diversity, supports short-chain fatty acid (SCFA) production, and strengthens the gut barrier.
🔬 The Science:
Whole grains contain resistant starch, beta-glucans, and arabinoxylans, which act as prebiotics, fuelling beneficial gut bacteria.
These fibres increase butyrate production, an SCFA that enhances gut barrier function, reduces inflammation, and promotes healthy immune regulation.
Studies have shown that individuals who consume fibre-rich whole grains have greater microbial diversity and a healthier gut lining than those on low-fibre, grain-free diets.
Yet, many practitioners still discourage grains, unintentionally starving their patients’ microbiomes of essential nutrients.
✅ Clinical Pearl: When addressing gut health, don’t just focus on eliminating "bad" foods—consider what’s missing. If a patient avoids grains, make sure they’re getting enough prebiotic fibre from other sources.
The Role of Grains in Gut Barrier Integrity
One of the biggest concerns around grains is their supposed link to “leaky gut.” But let’s take a step back—what really causes intestinal permeability?
✔️ Chronic stress
✔️ Dysbiosis
✔️ Poor diet (low fibre, high sugar, processed foods)
✔️ Alcohol, NSAIDs, and other gut-irritating substances
Meanwhile, whole grains contain key nutrients and compounds that actually help restore gut barrier function.
🔬 The Science:
Beta-glucans (found in oats and barley) have been shown to enhance mucosal immunity and support the gut lining.
Fermentable fibre from rye increases mucus production, providing a protective layer that prevents pathogenic bacteria from reaching the gut lining.
Inulin-rich grains (such as wheat and rye) promote the growth of beneficial bacteria which strengthen the intestinal barrier.
So why are we blaming grains for leaky gut? The real issue isn’t whole grains—it’s the absence of fibre in modern diets that’s leading to compromised gut health.
✅ Clinical Pearl: Before assuming grains are the cause of gut issues, look at the overall diet. If a patient is eating ultra-processed, refined grains, sugar, and low-fibre foods, that’s the real problem—not grains themselves.
Grains, Mucin-Degrading Bacteria, and Gut Resilience
Here’s something that isn’t talked about enough: low-fibre diets actually encourage bacteria to eat away at the gut lining.
When the gut doesn’t receive enough fermentable fibre (which whole grains provide), mucin-degrading bacteria start breaking down the intestinal mucus barrier for fuel, increasing susceptibility to pathogens and inflammation.
🔬 The Science:
In animal studies, a fibre-deficient diet led to increased mucus degradation, making the gut more vulnerable to infections and inflammation.
A human study showed that refined-grain diets increased mucin-degrading enzyme activity, whereas whole-grain diets promoted a healthier gut barrier and microbial balance.
This means avoiding grains can actually backfire—by depriving the gut of essential nutrients, we may be making it more vulnerable to inflammation and disease.
✅ Clinical Pearl: If a patient has gut issues and avoids grains, consider testing for microbial imbalances. A lack of fibre-feeding bacteria and an overgrowth of mucin-degrading bacteria could indicate that a low-grain diet is harming, rather than helping, their gut health.
The REAL Problem: Confusing Gluten Sensitivity with Other Gut Issues
One of the biggest mistakes in integrative medicine is blaming grains for symptoms that may actually be caused by other factors.
🚫 It’s not always gluten—it might be FODMAPs. Many people who believe they have gluten sensitivity are actually reacting to fructans (fermentable fibers found in wheat and rye), which are common IBS triggers.
🚫 It’s not always grains—it might be dysbiosis. If a patient experiences bloating or fatigue after eating grains, the real culprit could be SIBO, pathobiont overgrowth, or low microbial diversity.
🚫 It’s not always gluten—it might be food additives. Many processed gluten-containing foods are filled with preservatives, emulsifiers, and artificial ingredients that disrupt gut health—yet grains take the blame.
🚫 It’s not always grains—it might be how they’re prepared. Highly processed, refined grains have a very different impact on the body than properly soaked, sprouted, or fermented whole grains.
✅ Clinical Pearl: Instead of assuming grains are bad for a patient, consider their entire gut health picture—microbiome diversity, gut permeability, and overall diet quality matter far more than a single food category.
Step-by-Step Guide to Reintroducing Grains in Clinical Practice
For years, the health and wellness world has pushed the idea that removing grains is the solution to gut issues, inflammation, and metabolic dysfunction. But here’s the reality: for many patients, long-term grain avoidance is unnecessary—and in some cases, even harmful.
Why? Because grains—when chosen and prepared correctly—provide essential fermentable fibres, prebiotics, and nutrients that support microbiome diversity, gut barrier integrity, and metabolic health. If we want to move beyond outdated, fear-based nutrition, it’s time to get strategic about reintroducing grains rather than mindlessly eliminating them.
The process of reintroducing grains isn’t about throwing a loaf of bread at your patient and hoping for the best. It requires a structured, personalised approach that prioritises gut health, minimizes symptoms, and ensures long-term tolerance.
Here’s how to do it, step by step.
-
Before reintroducing grains, you need to understand why they were removed in the first place. Many patients avoid grains for reasons that have little to do with actual intolerance.
🔎 Key Questions to Ask:
✅ Was grain avoidance due to a medical diagnosis (e.g., coeliac, confirmed gluten sensitivity)?
✅ Did symptoms improve on a grain-free diet, or did they stay the same?
✅ What specific symptoms did grains cause in the past—bloating, fatigue, joint pain, brain fog?
✅ Has the patient’s gut health changed since eliminating grains (microbiome tests, stool analysis, symptom shifts)?
🚩 Red Flags That Require Extra Caution:
⚠️ Patients with a history of coeliac disease or wheat allergy should not reintroduce gluten.
⚠️ If the patient still experiences severe gut symptoms (e.g., chronic diarrhoea, bloating, histamine issues), optimize gut health before adding grains back in.
⚠️ If microbiome testing shows low microbial diversity or high mucin-degrading bacteria, fibre intake should be increased gradually to prevent gut stress.
✅ Clinical Pearl: Patients with past grain intolerance may have had an underlying dysbiosis, FODMAP sensitivity, or leaky gut—not a true grain issue. Fixing these before reintroduction is key.
-
Reintroducing grains does NOT mean adding back highly processed, refined carbs. We need to focus on nutrient-dense, fibre-rich grains that support gut health.
🥇 Best Grains for Reintroduction:
✔️ Quinoa – High in protein, fibre, and easy to digest.
✔️ Buckwheat – Naturally gluten-free and rich in polyphenols.
✔️ Millet – Gentle on digestion and packed with nutrients.
✔️ Sorghum – A powerful prebiotic that feeds beneficial gut bacteria.
✔️ Oats – Contain beta-glucans that support gut barrier function.
🥈 Next-Level Grains (Once Tolerance Is Established):
✔️ Rye – Excellent for microbiome diversity (contains arabinoxylans).
✔️ Barley – A powerhouse for short-chain fatty acid (SCFA) production.
✔️ Spelt – Lower in gluten, easier to digest than modern wheat.
🚫 Avoid These Initially:
❌ Highly processed white bread, cereals, and pasta – No fibre, no microbiome benefits.
❌ Instant oats and rice cakes – High glycaemic, low nutrient value.
❌ Commercial gluten-free products – Often contain emulsifiers, gums, and additives that can disrupt gut health.
✅ Clinical Pearl: Encourage whole, organic, and minimally processed grains—these will have the most microbiome-supportive benefits.
-
Many patients struggle with grains not because of the grains themselves, but because of how they’re processed and prepared. Traditional preparation methods reduce anti-nutrients (like phytic acid and lectins), enhance nutrient absorption, and improve digestibility.
🔬 Best Practices for Preparing Grains:
💧 Soaking – Soak grains (e.g., quinoa, buckwheat, oats) for 8-12 hours to reduce phytic acid and enhance digestion.
🌱 Sprouting – Sprouted grains are easier on the gut and contain higher nutrient levels.
🍞 Fermenting – True sourdough bread (fermented for 24+ hours) has lower gluten levels and a healthier impact on the microbiome.
🔥 Cooking with Bone Broth or Apple Cider Vinegar – Adding acidity or gelatine-rich broth to grains improves mineral absorption and digestion.
✅ Clinical Pearl: If a patient reacts poorly to grains, try changing the preparation method before assuming grains are the problem.
-
Reintroducing grains isn’t an all-or-nothing process. The gut microbiome needs time to adapt to new fibres and starches.
📏 Portion Guidelines for Reintroduction:
🔹 Start with 1-2 tablespoons of cooked grains per day.
🔹 Increase slowly to ½ cup over 3-6 months (depending on tolerance).
🔹 For highly reactive patients, begin with 1 teaspoon and track responses.
📅 Grain Reintroduction Schedule:
🗓️ Months 1-2 → Start with rice or buckwheat (small portions).
🗓️ Months 3-4 → Add in oats or millet (soaked overnight).
🗓️ Months 5-6 → Introduce rye or barley (properly prepared).
🗓️ Months 7+ → Expand to additional whole grains.
✅ Clinical Pearl: Symptoms like mild bloating or gas are normal during microbiome adaptation—but persistent issues (severe bloating, fatigue, loose stools) may indicate an underlying imbalance that needs addressing first.
-
The biggest mistake patients make when reintroducing grains? Eating the same grain every day. Repetitive exposure can increase sensitivity, while dietary diversity improves microbial balance and reduces intolerance risk.
🔄 Grain Rotation Guide:
✔️ Month 1 – Rice & buckwheat
✔️ Month 2 – Oats & millet
✔️ Month 3 – Sorghum & teff
✔️ Month 4 – Rye & barley
✅ Clinical Pearl: Encourage patients to rotate grains every few days/weeks to reduce overexposure and maximize nutrient variety.
-
Reintroducing grains is a dynamic process—it requires careful monitoring and adjustments.
📝 Track Symptoms Using a Food & Gut Journal:
📌 Digestive changes (bloating, gas, stool consistency)
📌 Energy levels (fatigue, brain fog, sluggishness)
📌 Skin reactions (eczema, acne, itching)
📌 Joint/muscle responses (stiffness, pain)
If a patient has negative reactions, don’t immediately assume “grains are bad.” Instead, adjust:
🔹 Try a different grain.
🔹 Modify the portion size.
🔹 Change the preparation method.
✅ Clinical Pearl: Symptoms may indicate gut dysbiosis, not grain intolerance. If issues persist, consider microbiome testing rather than blanket grain elimination.
-
Reintroducing grains isn’t just about adding them back—it’s about integrating them strategically into meals to support gut health, stabilize blood sugar, and enhance microbiome diversity.
🍽 Best Practices for Balanced Meals:
✅ Pair grains with protein – Helps stabilise blood sugar and improve satiety (e.g., quinoa + eggs, buckwheat + tempeh).
✅ Include healthy fats – Slows carbohydrate absorption and supports metabolism (e.g., oats + nuts, barley + avocado).
✅ Add fibre-rich foods – Enhances microbiome benefits (e.g., rye + lentils, sorghum + leafy greens).
✅ Incorporate fermented foods – Improves digestion and gut flora (e.g., sourdough + kimchi, oats + kefir).
🍽 Meal Ideas:
✔️ Breakfast: Soaked oats with berries, nuts, and yogurt.
✔️ Lunch: Quinoa and lentil salad with olive oil and fermented vegetables.
✔️ Dinner: Brown rice with roasted vegetables and salmon.
✅ Clinical Pearl: Encourage patients to rotate their grain choices and avoid eating the same grain daily. Variety supports microbial diversity and reduces the risk of intolerance.
Microbiome Testing and Grains
Microbiome testing provides invaluable insights into the diversity and composition of your patient’s gut microbiome, allowing you to make more precise decisions when deciding if grains should or shouldn’t be included in their treatment plan. Understanding which microbial species are abundant or lacking helps you tailor prescriptions to each patient’s unique microbiome, optimising therapeutic outcomes.
By integrating microbiome testing, such as the Microbiome Explorer by Microba, into your clinical practice, you can personalise polyphenol interventions based on robust, data-driven insights. This approach ensures that you maximize the therapeutic potential of polyphenols in improving gut health and addressing your patients’ specific clinical needs.
Conclusion: Rethinking Grains – A Balanced, Science-Based Approach
For years, grains have been unfairly demonised in the world of nutrition, labelled as gut-disrupting, inflammatory, and even unnecessary. But as we’ve explored, the real issue is not grains themselves—it’s how they are chosen, prepared, and consumed.
We’ve unpacked the common myths and provided a structured, science-backed approach for reintroducing them in clinical practice. The key takeaway? Precision over restriction.
✔️ Whole grains—rich in fibre, resistant starch, and polyphenols—feed the microbiome, promote short-chain fatty acid production, and strengthen gut barrier integrity.
✔️ Refined grains—stripped of their nutrients—offer little benefit and can contribute to metabolic dysfunction.
✔️ Gluten is not the universal villain it has been made out to be; for many patients, other gut imbalances (dysbiosis, FODMAP intolerance, ultra-processed foods) are the real culprits.
✔️ Practitioners must shift from fear-based nutrition to evidence-based decision-making—instead of eliminating grains by default, we should be testing, assessing, and tailoring recommendations to the individual.
What This Means for Clinical Practice
The best practitioners don’t follow dietary trends—they challenge them. Moving forward:
✅ Question Assumptions – Don’t recommend grain-free diets without a clear, clinical reason for doing so.
✅ Use Science, Not Fear – Research supports the role of properly prepared whole grains in gut health.
✅ Tailor Nutrition to the Individual – Some patients thrive on grains, while others may need modifications—but the key is personalisation, not blanket restriction.
✅ Educate Patients – Many people avoid grains based on misinformation. Help them understand which grains, in what form, and why they may or may not be beneficial.
Grains do not need to be the enemy—they can be a powerful tool for gut health when used correctly. Instead of choosing elimination by default, let’s embrace precision nutrition and make decisions based on science, not fear.
Dr Brad Leech
Brad is a PhD-qualified Clinical Nutritionist and Herbalist specialising in chronic autoimmune conditions and complex gastrointestinal disorders. He provides complete and personalised care to his patients using functional nutrition, integrative medicine and holistic wellness.