The Ultimate Guide to Managing Autoimmune Diseases with Integrative and Functional Medicine
Why an Integrative Medicine Approach is Key to Autoimmune Disease Management
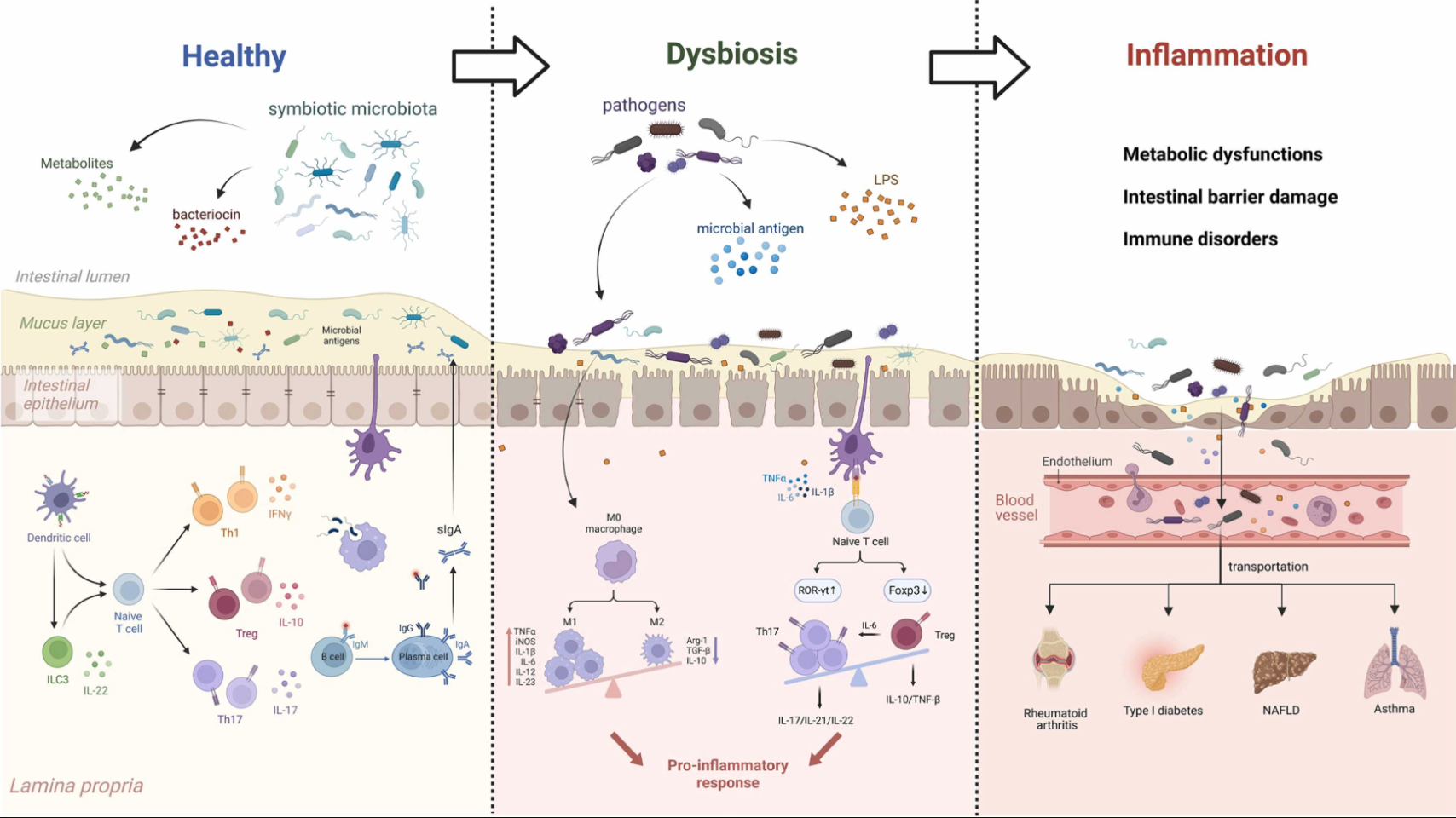
Autoimmune diseases represent a growing global health challenge, with their prevalence increasing at an alarming rate across diverse populations. Characterised by a loss of immune tolerance and the subsequent attack on self-tissues, autoimmune diseases manifest through a wide spectrum of clinical presentations, from systemic inflammation in rheumatoid arthritis to organ-specific damage in conditions such as type 1 diabetes and celiac disease. The complexity of autoimmune diseases pathogenesis is driven by an intricate interplay of genetic susceptibility, environmental triggers, and dysregulated immune responses. Yet, the role of modifiable factors—diet, lifestyle, and the gut microbiome—has emerged as a pivotal axis in both the onset and progression of these conditions.
Among the modifiable risk factors, the health of the gut and its associated microbiome stands as a cornerstone in the management of autoimmune disease. The gut is not merely a digestive organ; it functions as an immunological command centre, housing nearly 70% of the body’s immune cells. Dysbiosis, a state of microbial imbalance, and increased intestinal permeability ("leaky gut") have been identified as key drivers of chronic inflammation and immune dysregulation, both of which are hallmarks of autoimmunity. In clinical practice, this makes the gut an indispensable target for therapeutic interventions aimed at modulating systemic immune responses.
This article serves as an advanced guide, offering a deep dive into evidence-based dietary and lifestyle interventions to manage autoimmune diseases. By exploring the latest research and clinical insights, you will gain a comprehensive understanding of the interplay between gut health, systemic inflammation, and immune modulation. Through this guide, I aim to transform the way autoimmune diseases are managed, bringing the principles of functional nutrition, microbiome science, and integrative medicine to the forefront of patient care. The information presented in this article has been taken from my own published research.
Take Your Expertise to the Next Level Join the NEW Autoimmune Mentoring Program
Are you ready to expand your knowledge, refine your clinical skills, and integrate the latest microbiome research into your practice?
📢 Join My NEW Brad’s Brainiacs: Autoimmune Mentoring Program!
This mentorship program is designed specifically for practitioners who want to deepen their understanding of autoimmune disease and the microbiome. Whether you're a naturopath, nutritionist, functional medicine doctor, or integrative health professional, this program will provide you with the tools and confidence to implement cutting-edge, evidence-based strategies in your clinic.
🔹 What you’ll gain:
✔️ The latest scientific insights into how the microbiome influences autoimmunity.
✔️ A structured approach to using functional testing like metagenomic sequencing to guide treatment decisions.
✔️ Practical clinical protocols for gut healing, immune modulation, and patient-specific interventions.
✔️ Exclusive case studies and real-world applications to help you apply these insights with confidence.
✔️ Access to a community of like-minded practitioners, sharing experiences, case discussions, and the latest research.
Understanding the Root Causes of Autoimmune Diseases – Gut Health, Dysbiosis, and Chronic Inflammation
Autoimmune diseases stem from a dynamic and multifactorial interaction between genetic predisposition, environmental triggers, and dysregulated immune function. While these conditions exhibit diverse clinical manifestations, they share common underlying pathophysiological mechanisms: gut dysbiosis, increased intestinal permeability, chronic inflammation and immune dysfunction. Understanding these mechanisms is critical to target the root causes of autoimmune diseases and restore immune tolerance.
Dysbiosis: A Catalyst for Immune Dysregulation
The gut microbiome, a dense and metabolically active community of microorganisms, orchestrates a multitude of immune processes. It produces short-chain fatty acids (SCFAs), such as butyrate, which maintain intestinal barrier integrity and modulate systemic inflammation. Dysbiosis—defined as an imbalance in microbial composition—undermines these protective mechanisms, creating an environment conducive to autoimmunity.
Key mechanisms through which dysbiosis contributes to autoimmune diseases pathogenesis include:
Reduction in Microbial Diversity: Loss of keystone species, such as Faecalibacterium prausnitzii, reduces SCFA production, impairing gut barrier function and promoting inflammation.
Overgrowth of Pathobionts: Pathobionts, such as Bilophila wadsworthia, thrive on diets rich in long-chain saturated fats, triggering local and systemic inflammatory responses via toll-like receptor (TLR) activation.
Microbial Translocation: Dysbiosis increases intestinal permeability, enabling microbial antigens, such as lipopolysaccharides (LPS), to translocate into the systemic circulation, thereby amplifying chronic inflammation.
Dysbiosis is not merely a consequence of autoimmune diseases but an active driver, perpetuating the dysregulated immune responses that define these conditions. Correcting dysbiosis through dietary interventions and prebiotics becomes a cornerstone in modulating autoimmune diseases pathophysiology.
Increased Intestinal Permeability: The "Leaky Gut" Phenomenon
The intestinal epithelial barrier is a critical structure that protects the immune system from undue exposure to luminal antigens. Composed of a single layer of epithelial cells, its integrity is regulated by tight junction proteins, such as zonulin. Disruption of these tight junctions leads to increased intestinal permeability, often referred to as “leaky gut.”
Increased permeability allows luminal contents, including dietary antigens, microbial components, and toxins, to stimulate the intestinal barrier and interact with the underlying immune system. This process triggers:
Activation of the Innate Immune System: Translocated antigens, such as LPS, activate pattern recognition receptors (e.g., TLR4), leading to the production of pro-inflammatory cytokines like IL-6 and TNF-α.
Molecular Mimicry: Certain dietary and microbial antigens mimic host tissues, leading to autoreactive T-cell activation and the development of autoimmunity. Gluten, for example, may promoting autoimmunity through molecular mimicry.
Recent evidence suggests that even individuals without overt intestinal symptoms may exhibit subclinical levels of increased intestinal permeability, linking this phenomenon to the early stages of autoimmune disease development. Interventions that restore barrier integrity, such as dietary modifications and butyrate-enhancing therapies, are therefore integral to autoimmune disease management.
Chronic Inflammation: The Immune System in Overdrive
Chronic low-grade inflammation underpins the development and progression of autoimmune diseases. This persistent inflammatory state results from the interplay of dysbiosis, increased permeability, and immune dysregulation.
Key players in chronic inflammation include:
Cytokine Imbalance: Pro-inflammatory cytokines, such as IL-17, IL-23, and TNF-α, dominate the immune landscape in ADs. These cytokines exacerbate tissue damage and impair regulatory T-cell (Treg) function, which is critical for maintaining self-tolerance.
Th17 Cell Polarisation: Environmental and dietary factors, such as excessive sodium intake or dysbiosis, promote the differentiation of Th17 cells. These cells secrete IL-17, driving autoimmune processes and perpetuating inflammation.
NF-κB Pathway Activation: Chronic activation of nuclear factor-kappa B (NF-κB) by microbial antigens and dietary triggers amplifies the inflammatory cascade, further damaging tissues and organs.
Notably, chronic inflammation is not confined to the site of immune attack but also contributes to systemic comorbidities, including cardiovascular disease and metabolic dysfunction. Integrative strategies that modulate inflammatory pathways, such as omega-3 fatty acids, curcumin, and anti-inflammatory diets, are pivotal in breaking this cycle.
Clinical Implications for Practitioners
Understanding the core pathophysiology of autoimmune diseases helps to understand the root causes of immune dysregulation. Key takeaways include:
Dysbiosis Correction: Implementing microbiome-focused interventions, such as prebiotics, probiotics, and dietary fibre, to restore microbial diversity.
Barrier Restoration: Enhancing intestinal integrity through SCFA production, zonulin modulation, and elimination of dietary triggers.
Inflammation Mitigation: Reducing systemic inflammation via anti-inflammatory diets, targeted supplementation, and lifestyle modifications.
By targeting these interconnected mechanisms, practitioners can move beyond symptom management to achieve true immune homeostasis and significantly improve patient outcomes in autoimmune disease care.
Modifiable Lifestyle and Dietary Factors that Trigger or Heal Autoimmune Diseases
Autoimmune diseases are driven by an interplay of genetic susceptibility, environmental exposures, and immune dysregulation. While genetic predisposition is immutable, modifiable factors such as diet, lifestyle, and environmental influences play a central role in shaping the immune response and disease trajectory. Targeting these modifiable risk factors offers a powerful approach to mitigating the development and progression of autoimmune diseases.
Diet: A Central Driver of Autoimmune Pathogenesis
Dietary patterns are among the most influential modifiable factors in autoimmune disease, exerting direct effects on the gut microbiota, intestinal barrier, and systemic inflammation. Modern dietary habits, characterised by high intakes of processed foods, sugar, and unhealthy fats, promote pro-inflammatory states, dysbiosis, and immune dysregulation.
Key dietary contributors to autoimmune risk include:
Long-Chain Saturated Fats (LCSFs): These fats, abundant in animal-derived foods, activate Toll-like receptor 4 (TLR4), promoting nuclear factor-kappa B (NF-κB) activity and downstream pro-inflammatory cytokines such as IL-6 and TNF-α. Chronic consumption of LCSFs exacerbates dysbiosis and increases intestinal permeability, perpetuating autoimmune processes.
Arachidonic Acid: Found predominantly in animal products, arachidonic acid is a precursor to pro-inflammatory eicosanoids such as prostaglandins and leukotrienes. Diets rich in arachidonic acid (>90 mg/day) correlate with heightened inflammation and disease activity in conditions like rheumatoid arthritis.
Conversely, anti-inflammatory dietary components can mitigate these risks:
Omega-3 Polyunsaturated Fatty Acids (PUFAs): EPA and DHA from fatty fish suppress inflammatory mediators, shift Th17/Treg cell balance, and support intestinal integrity.
Fibre and Prebiotics: High-fibre foods enhance microbial production of short-chain fatty acids (SCFAs), particularly butyrate, which fortifies the gut barrier and reduces inflammation.
Polyphenols: Antioxidant compounds found in fruits, vegetables, and teas reduce oxidative stress, modulate immune pathways, and support microbiota diversity.
Dietary intervention is not merely palliative but transformative. Tailored nutritional strategies that eliminate inflammatory triggers, support gut health, and optimise nutrient intake form the foundation of an effective autoimmune disease management plan.
Lifestyle Factors: The Impact of Behaviour on Immune Regulation
Chronic stress, physical inactivity, and poor sleep hygiene are powerful contributors to autoimmune disease via their effects on the hypothalamic-pituitary-adrenal (HPA) axis, systemic inflammation, and immune regulation.
Stress and the HPA Axis
Chronic psychological stress disrupts HPA axis homeostasis, increasing circulating cortisol levels and impairing the immune system's ability to regulate inflammation. Elevated cortisol suppresses Treg cells and enhances Th17-mediated immune responses, driving autoimmune activity. Stress management techniques such as mindfulness-based stress reduction (MBSR), yoga, and meditation can significantly reduce inflammatory biomarkers and improve clinical outcomes in AD patients.Physical Activity
Sedentary lifestyles are associated with increased systemic inflammation, insulin resistance, and dysbiosis. Moderate, regular exercise improves Treg cell function, reduces inflammatory cytokines, and enhances gut microbiota diversity. Activities such as brisk walking, swimming, or low-impact resistance training are particularly beneficial in autoimmune populations.Sleep Quality
Sleep deprivation increases inflammatory markers (e.g., IL-6, CRP) and disrupts immune homeostasis. Poor sleep exacerbates autoimmune conditions by impairing the circadian regulation of immune responses. Addressing sleep hygiene, including consistent sleep schedules, limiting screen time before bed, and managing sleep disorders, is essential for improving immune regulation in ADs.
Environmental Exposures: Hidden Triggers of Autoimmunity
Environmental toxins, infections, and food additives have profound implications for autoimmune disease risk. These exposures often act as triggers in genetically predisposed individuals, initiating or exacerbating disease processes.
Pollutants and Toxins
Heavy metals (e.g., mercury, lead), pesticides, and industrial chemicals disrupt immune homeostasis through oxidative stress, mitochondrial dysfunction, and T-cell dysregulation. Integrative detoxification strategies, including chelation therapy (for specific toxins) and antioxidant support (e.g., glutathione, NAC), can help mitigate these effects.Infections and Molecular Mimicry
Certain infections, such as Epstein-Barr virus and Helicobacter pylori, are associated with autoimmune diseases through molecular mimicry. These pathogens produce antigens that resemble host tissues, triggering cross-reactive immune responses. Supporting the immune system with antivirals, immune-modulating herbs (e.g., echinacea, astragalus), and targeted probiotics can reduce infectious burden and immune dysregulation.Food Additives
Emulsifiers, artificial sweeteners, and nanoparticles disrupt the gut microbiome and increase intestinal permeability, amplifying inflammation and autoimmune risk. Avoiding processed foods and prioritising whole, minimally processed ingredients is a critical component of any autoimmune management plan.
Clinical Implications for Practitioners
Targeting modifiable risk factors may influence the trajectory of autoimmune diseases. Key strategies include:
Dietary Optimisation: Eliminate pro-inflammatory foods, incorporate anti-inflammatory and microbiome-supportive nutrients, and personalise interventions based on patient-specific triggers.
Stress and Sleep Management: Integrate stress reduction techniques and promote restorative sleep to modulate immune responses.
Environmental Detoxification: Identify and mitigate environmental exposures, using both lifestyle adjustments and targeted therapies.
Promoting Physical Activity: Recommend appropriate exercise tailored to the patient’s physical capacity and disease state.
By addressing these interconnected risk factors, practitioners can shift the focus from disease suppression to restoration of immune homeostasis, empowering patients to take control of their health and reducing the burden of autoimmune disease.
Best Anti-Inflammatory Diets for Autoimmune Diseases – Evidence-Based Nutrition Plans
Dietary interventions have emerged as a cornerstone in managing autoimmune diseases, directly influencing the gut microbiome, intestinal barrier integrity, and systemic immune responses. The integrative medicine approach seeks to individualise these interventions, focusing on both removing harmful dietary triggers and incorporating nutrient-dense, anti-inflammatory foods to restore immune homeostasis.
Critical Evaluation of Popular Autoimmune Diets
Gluten-Free Diet
A gluten-free diet is indispensable for individuals with celiac disease and often beneficial in non-celiac autoimmune conditions due to gluten's role in increasing intestinal permeability and triggering molecular mimicry. Many patients report symptom improvement with gluten elimination, even in conditions such as Hashimoto’s thyroiditis and RA.
Limitations:
Gluten-free processed foods often lack essential nutrients and may be high in refined carbohydrates, which can perpetuate inflammation.
For patients without confirmed gluten sensitivity, strict adherence may unnecessarily limit dietary diversity.
Clinical Perspective: While universally necessary for celiac disease, a gluten-free diet should be personalised for other autoimmune conditions, prioritising whole, nutrient-dense gluten-free options over processed substitutes.
Paleolithic (Paleo) Diet
The Paleo diet eliminates modern dietary triggers, including grains, dairy, legumes, and processed foods, focusing instead on lean proteins, fruits, vegetables, and nuts. This diet aligns with the need to reduce inflammatory triggers and support gut health.
Limitations:
The exclusion of legumes and grains may lead to suboptimal fibre intake, potentially limiting microbiome diversity.
Without careful planning, calcium and vitamin D intake may be insufficient due to the exclusion of dairy.
Clinical Perspective: The Paleo diet provides a useful template for autoimmune disease management but requires adjustments to ensure adequate fibre and micronutrient intake.
Low-Arachidonic Acid Diet
A diet restricting arachidonic acid (<90 mg/day) focuses on reducing pro-inflammatory precursors derived from animal products. This approach has shown efficacy in conditions like RA by decreasing inflammatory eicosanoids.
Limitations:
Over-restriction of animal products may lead to inadequate protein intake, particularly in patients with higher metabolic demands.
Practicality can be a concern, as it necessitates significant dietary tracking.
Clinical Perspective: While beneficial for highly inflammatory states, this diet should be combined with strategies to maintain balanced protein and essential nutrient intake.
Synthesising the Evidence: The Most Effective Approach
Given the strengths and limitations of individual diets, a hybrid approach that integrates their evidence-based elements provides the most effective framework for practitioners. This personalised strategy is designed to combine the strengths of various dietary protocols while minimising their shortcomings.
Core Principles of the Autoimmune-Friendly Whole-Food Diet:
Eliminate Known Triggers:
Remove gluten and other common dietary antigens during the initial phases of treatment to reduce immune activation.
Focus on Anti-Inflammatory Foods:
Prioritise omega-3-rich fish, colourful fruits and vegetables (6+ servings daily), and polyphenol-dense foods such as green tea, turmeric, and berries.
Incorporate Gut-Supportive Foods:
Include fibre-rich vegetables, prebiotics (e.g., asparagus, garlic), and fermented foods (e.g., sauerkraut, kefir) to enhance microbiota diversity.
Tailor Macronutrient Ratios:
Adjust carbohydrate, fat, and protein proportions based on the patient’s unique needs, focusing on low-glycaemic-load options and high-quality proteins.
Emphasise Whole, Minimally Processed Foods:
Avoid processed gluten-free or "Paleo" substitutes that lack nutrients or include additives. Opt for whole, nutrient-dense options like sweet potatoes, quinoa, and organic lean proteins.
Supplements and Functional Foods That Transform Autoimmune Disease Management
While dietary changes form the cornerstone of autoimmune disease management, targeted supplementation and functional foods offer additional therapeutic opportunities. By addressing specific nutrient deficiencies, modulating the gut microbiome, and attenuating systemic inflammation, supplements and functional foods enhance the efficacy of dietary interventions.
Probiotics: Restoring Microbial Balance
Dysbiosis plays a central role in autoimmune disease pathogenesis, making probiotics a valuable adjunct to dietary interventions. These live microorganisms modulate gut microbiota composition, enhance intestinal barrier function, and regulate immune responses.
Key mechanisms of action include:
Restoration of Microbiota Diversity: Probiotics promote the growth of beneficial bacteria, which support SCFA production and gut integrity.
Regulation of Immune Responses: Certain strains, such as Lactobacillus rhamnosus GG, reduce Th17-mediated inflammation and promote Treg activity.
Reduction in Intestinal Permeability: Probiotics strengthen tight junctions, reducing antigen translocation and systemic immune activation.
Clinical Tip: Use strain-specific probiotics for targeted outcomes, and consider prebiotic supplementation to enhance their efficacy.
Omega-3 Fatty Acids: Master Regulators of Inflammation
Omega-3 polyunsaturated fatty acids (PUFAs), particularly EPA and DHA, are among the most effective natural agents for reducing inflammation. These essential fatty acids modulate both innate and adaptive immune responses, making them invaluable in autoimmune disease management.
Key benefits include:
Eicosanoid Modulation: Omega-3s suppress the production of pro-inflammatory eicosanoids (e.g., prostaglandins and leukotrienes) derived from arachidonic acid.
Th17/Treg Balance: EPA and DHA reduce Th17 cell activity and enhance Treg cell function, restoring immune tolerance.
Intestinal Barrier Integrity: Omega-3s improve tight junction function and decrease intestinal permeability.
Clinical trials demonstrate that omega-3 supplementation reduces disease activity in rheumatoid arthritis, multiple sclerosis, and systemic lupus erythematosus. A typical therapeutic dose ranges from 2 to 4 grams of EPA/DHA daily.
Clinical Tip: Choose high-quality, purified fish oil supplements to minimise contaminants like mercury and PCBs.
Vitamin D: Modulating Immune Tolerance
Vitamin D plays a critical role in immune regulation, influencing both innate and adaptive immunity. Its deficiency is strongly associated with increased risk and severity of autoimmune diseases.
Mechanisms of action include:
Treg Activation: Vitamin D enhances Treg function, promoting self-tolerance and reducing autoimmune activation.
Suppression of Inflammatory Cytokines: Vitamin D inhibits IL-17 and TNF-α production, shifting the immune response away from a pro-inflammatory phenotype.
Barrier Function: Vitamin D supports the maintenance of tight junction integrity, reducing intestinal permeability.
Clinical studies indicate that correcting vitamin D deficiency improves disease outcomes in conditions such as multiple sclerosis, type 1 diabetes, and systemic lupus erythematosus. Serum 25(OH)D levels should be maintained at 100–125 nmol/L for optimal immune health.
Clinical Tip: Test patients' vitamin D levels and tailor dosing, typically 2,000–5,000 IU/day, to achieve optimal serum concentrations.
Polyphenols: Potent Anti-Inflammatory Agents
Polyphenols, bioactive compounds found in plant-based foods, exert profound antioxidant, anti-inflammatory, and immune-modulating effects. Key sources include green tea, turmeric, berries, and pomegranate.
Key actions include:
NF-κB Suppression: Polyphenols inhibit the activation of nuclear factor-kappa B (NF-κB), a key driver of inflammation in autoimmune diseases.
Gut Microbiome Modulation: Polyphenols promote the growth of beneficial gut bacteria, enhancing SCFA production and microbial diversity.
Regulation of Oxidative Stress: By neutralising reactive oxygen species (ROS), polyphenols prevent oxidative damage to tissues and cells.
Curcumin, a polyphenol derived from turmeric, has shown particular promise in autoimmune conditions. It reduces inflammatory cytokines (e.g., TNF-α, IL-6) and modulates immune cell activity. Clinical studies support curcumin's use in RA, ulcerative colitis, and psoriasis.
Clinical Tip: Use bioavailable forms of curcumin (e.g., those combined with piperine or phospholipids) to enhance absorption and therapeutic effect.
A Practical Guide to Personalising Autoimmune Treatment Plans Patients
Managing autoimmune diseases in clinical practice requires a patient-centred, multifaceted approach.We must balance advanced clinical insights with practical strategies that empower patients to implement meaningful lifestyle changes. This section provides a structured framework to help clinicians optimise outcomes through personalised care plans, functional testing, and strategies for improving adherence.
Personalising Nutrition Plans for Autoimmune Patients
Autoimmune diseases are heterogeneous in their clinical presentations and triggers, making personalised nutrition central to successful management.
Steps to Personalisation:
Identify Triggers Through Elimination Diets:
Use a structured elimination and reintroduction approach to identify dietary antigens that exacerbate symptoms. Begin with eliminating common triggers for 4–6 weeks and monitor symptom improvement.
Tailor Diets Based on Specific Conditions:
Celiac Disease: Strict gluten elimination is essential. Incorporate nutrient-dense gluten-free grains like quinoa and amaranth.
Rheumatoid Arthritis: Focus on a low-arachidonic acid diet supplemented with omega-3s and polyphenol-rich foods like turmeric and green tea.
Inflammatory Bowel Disease: Emphasise low-fibre diets during flares and gradually transition to high-fibre, SCFA-promoting diets in remission.
Clinical Tip: Incorporate patient preferences and cultural dietary patterns to ensure adherence and sustainability.
Leveraging Functional Testing to Guide Interventions
Functional testing offers valuable insights into the root causes and drivers of autoimmunity, enabling practitioners to design precise interventions.
Key Tests and Their Applications:
Microbiome Testing:
Assess microbial diversity, dysbiosis markers, and SCFA production to tailor prebiotic and probiotic strategies.
Identify overgrowth of pathobionts (e.g., Bilophila wadsworthia) linked to inflammation.
Intestinal Permeability Assessment:
Measure stool zonulin and Hexa-LPS
Elevated markers guide the use of interventions like glutamine, butyrate, and zinc carnosine to restore barrier integrity.
Blood pathology:
Evaluate levels of vitamin D, inflammation, and other key nutrients(e.g., selenium, zinc, B vitamins). Correcting deficiencies directly influences immune regulation and inflammation.
I've hand selected the most important blood markers in Dr Brad Leech: Optimal Health Check
Clinical Tip: Combine testing results with clinical history and symptoms to avoid over-reliance on biomarkers alone.
Implementing Lifestyle Modifications for Immune Regulation
Beyond diet, lifestyle factors profoundly impact immune balance. Integrative strategies must address stress, sleep, and physical activity.
Stress Management:
Chronic stress perpetuates immune dysregulation via hypothalamic-pituitary-adrenal (HPA) axis dysfunction. Techniques such as mindfulness-based stress reduction (MBSR), yoga, and guided imagery lower cortisol levels and improve Treg cell function.
Encourage daily practices, such as 10 minutes of deep breathing or gratitude journaling, to build resilience against stressors.
Improving Sleep Hygiene:
Sleep deprivation exacerbates inflammation through upregulation of IL-6 and TNF-α.
Recommend consistent sleep schedules, blue light reduction, and herbal sleep aids like valerian or magnesium glycinate to optimise restorative sleep.
Incorporating Exercise:
Moderate-intensity aerobic exercise and resistance training reduce systemic inflammation and enhance microbiota diversity.
Tailor activity levels to patient capacity, particularly during disease flares, to avoid overexertion.
Clinical Tip: Use wearable devices to monitor stress, sleep, and activity levels, fostering patient accountability and progress tracking.
Enhancing Patient Adherence with Behavioural Strategies
Behavioural barriers, such as overwhelm or perceived complexity, often hinder adherence to treatment plans. Practitioners must employ evidence-based strategies to drive sustainable change.
The "Atomic Habits" Approach to Building Healthy Behaviours:
Make It Simple: Break dietary and lifestyle changes into manageable steps. For example, start with eliminating one trigger food (e.g., gluten) rather than overhauling the entire diet immediately.
Stack New Habits with Existing Routines: Suggest pairing new habits with established ones. For instance, "Take your probiotics after brushing your teeth in the morning."
Use Visual Cues: Recommend patients keep healthy snacks visible and store supplements in accessible places to reinforce consistency.
Track Progress: Encourage patients to log meals, symptoms, and lifestyle habits to identify patterns and celebrate small wins.
Patient Empowerment:
Educate patients on the "why" behind recommendations, linking interventions to specific mechanisms of disease modulation (e.g., “Reducing red meat can restore gut barrier integrity, reducing systemic inflammation.”).
Provide resources such as meal plans, recipes, and mindfulness apps to make changes accessible and achievable.
Clinical Tip: Schedule regular follow-ups to monitor adherence, address challenges, and adjust plans as needed.
Managing Your Autoimmune Disease: A Patients Guide to Wellness
About the presentation:
In this 45min presentation, Dr Brad Leech will discuss the intricate world of autoimmune conditions, delving into their root causes and modifiable risk factors. He will shed light on the connections between leaky gut, dysbiosis, inflammation, and immune dysregulation, offering insights into measuring these factors. Dr Brad Leech will present holistic and integrative strategies using diet, lifestyle, and supplements to address these issues and improve your autoimmune health.
Conclusion: Integrative Medicine as the Future of Autoimmune Disease Care
Autoimmune diseases demand a root-cause approach, integrating personalised nutrition, lifestyle optimisation, and functional interventions. By addressing the core drivers of autoimmunity—dysbiosis, intestinal permeability, and chronic inflammation— we can move beyond symptom management to achieve meaningful disease modulation.
As practitioners, we must leverage the tools of functional testing to identify individualised risk factors, monitor progress, and refine interventions. Personalised care is not a luxury in autoimmune disease management; it is a necessity. This precision-driven approach allows us to intervene at the level of underlying mechanisms, offering patients the opportunity for true disease modulation rather than temporary remission.
The future of autoimmune disease management lies in the integrative model. By synthesising the best of evidence-based nutrition, functional medicine, and patient-centred care, we have the potential to shift the paradigm from reactive to proactive healthcare. This approach not only alleviates disease burden but also restores hope—empowering patients to reclaim their health and live with greater vitality.
Dr Brad Leech
Brad is a PhD-qualified Clinical Nutritionist and Herbalist specialising in chronic autoimmune conditions and complex gastrointestinal disorders. He provides complete and personalised care to his patients using functional nutrition, integrative medicine and holistic wellness.